 Another possibility is a carbon with three bonds and a single, unpaired (free radical) electron: in this case, the carbon has a formal charge of zero. If, on the other hand, it has three bonds plus a lone pair of electrons, it will have a formal charge of -1. The next example further demonstrates how to calculate formal charges. In cases where there are positive or negative formal charges on various atoms, stable structures generally have negative formal charges on the more electronegative atoms and positive formal charges on the less electronegative atoms. Typically, the structure with the most charges on the atoms closest to zero is the more stable Lewis structure. When summed the overall charge is zero, which is consistent with the overall charge on the NH 3 molecule. In this example, the nitrogen and each hydrogen has a formal charge of zero. 
Adding together the formal charges should give us the overall charge on the molecule or ion. The hydrogen atoms in ammonia have the same number of electrons as neutral hydrogen atoms, and so their formal charge is also zero. Using Equation 4.4.1 to calculate the formal charge on hydrogen, we obtain:įormalcharge(H)=1 valence e −−(0 non−bonding e − + 2 bonding e −/2)=0 Each hydrogen atom in the molecule shares one pair of bonding electrons and is therefore assigned one electron. Substituting into the formula, we obtainįormalcharge(N)=5 valence e −−(2 non−bonding e − + 6 bonding e −/2)=0Ī neutral hydrogen atom has one valence electron. From its Lewis electron structure, the nitrogen atom in ammonia has one lone pair and shares three bonding pairs with hydrogen atoms, so nitrogen itself is assigned a total of five electrons. To illustrate this method, let’s calculate the formal charge on the atoms in ammonia (NH 3) whose Lewis electron structure is as follows:Ī neutral nitrogen atom has five valence electrons (it is in group 15). Bonding electrons are divided equally between the bonded atoms.įor each atom, we then compute a formal charge:įormal charge = valence e − −(free atom)(non−bonding e − + bonding e – /2). 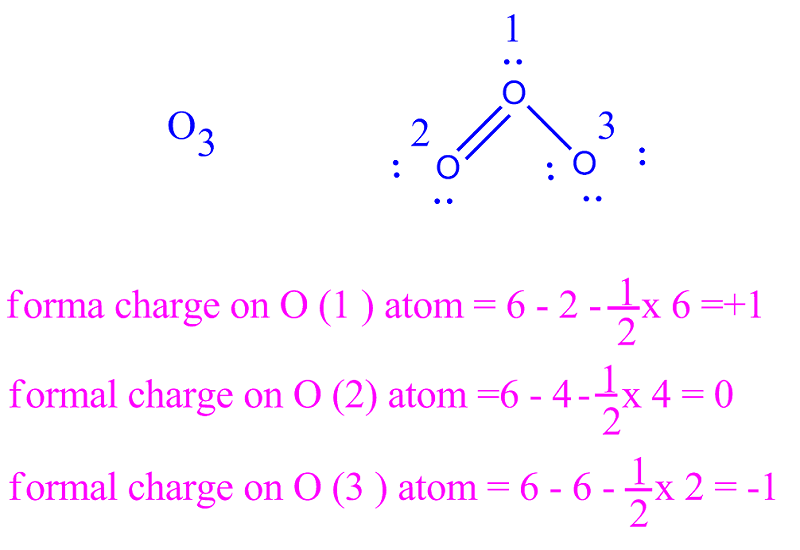
Nonbonding electrons are assigned to the atom on which they are located. 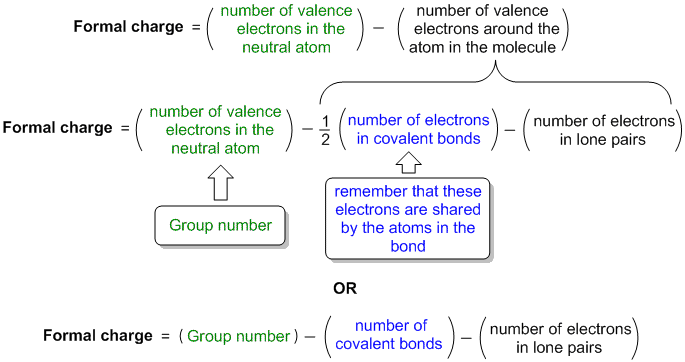
To calculate formal charges, we assign electrons in the molecule to individual atoms according to these rules: A formal charge does not represent a true charge on an atom in a covalent bond but is simply used to predict the most likely structure when a compound has more than one valid Lewis structure. The formal charge is a way of computing the charge distribution within a Lewis structure the sum of the formal charges on the atoms within a molecule or an ion must equal the overall charge on the molecule or ion. In these situations, we can choose the most stable Lewis structure by considering the formal charge on the atoms, which is the difference between the number of valence electrons in the free atom and the number assigned to it in the Lewis electron structure. It is sometimes possible to write more than one Lewis structure for a substance that does not violate the octet rule, as we saw for CH 2O, but not every Lewis structure may be equally reasonable. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
